Fatty acids
Fatty acids have a chemical structure with: of course a carboxylic group (they are indeed acids) + a carbon chain of about 15 to 20 C atoms.
Mostly the fatty acids occur in the compounds with glycerine, but now and then they occur freely (for example, stearic acid = candle, C17H35COOH).
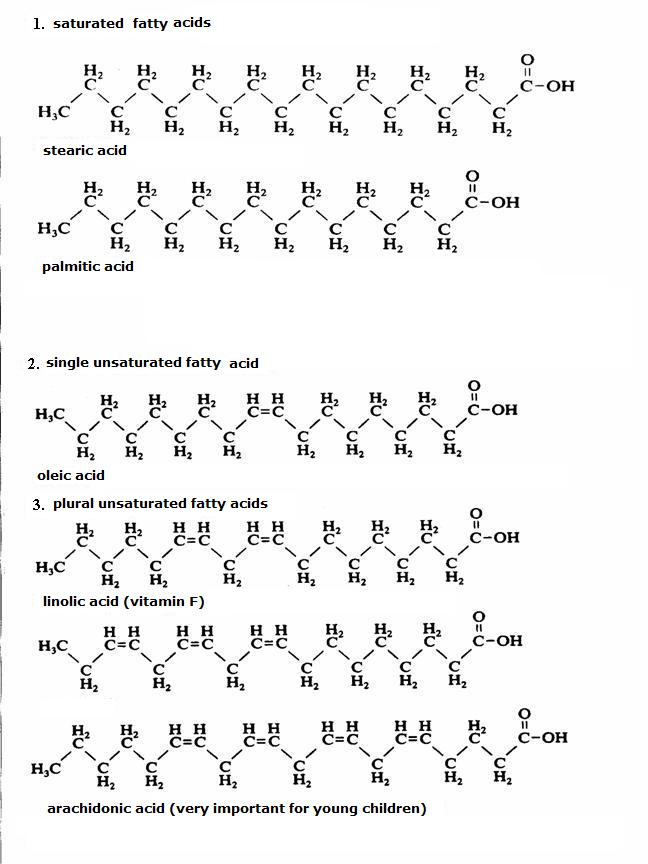
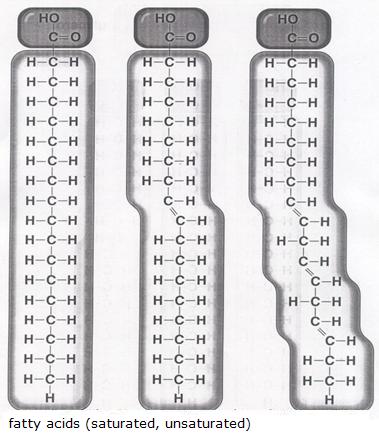
In the figure you see the structures of three fatty acids.
One of them in stearic acid (candle); the other has a double bond, so is unsaturated; the third has several double bonds, so is plurally unsaturated.
The next image shows four models: three models of unsaturated fatty acid + one mode of glycerine.
The three fatty acids react, each of them with one OH-group of the glycerine, under simultaneously formation of three molecules of water.

And in the next image you can see the products: a fatty acid + the three produced water molecules:




