Mono-, di-, (oligo-) an polysaccharides
Examples:
- glucose and fructose
- saccharose and maltose
- amylose and cellulose in plants, and glycogene in animals
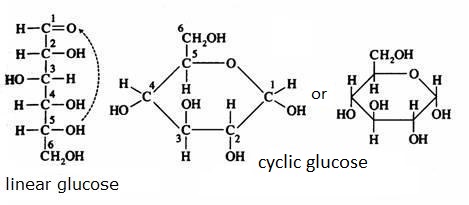
Control the formula C6H12O6 in the images below:


models of the cyclic structure of glucose
Monosaccharides
They are the monomers of the polysaccharides. There are various types of monosaccharides, depending on:
- the number of C atoms: tetrose, pentose, hexose, heptose...
- the presence of an aldehyde or a keton group: aldose and ketose
- the structure of the whole molecules, linear or open, or: closed or cyclic.
five examples of monosaccharides:
Glucose α and β
Galactose
Fructose
Ribose
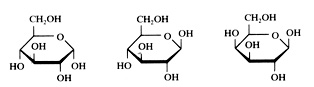
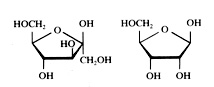
Cyclic or linear
Monomers normally have both structures: cyclic and linear. The two are continuously in equilibrium with each other (supposed that they are dissolved in water).
Note that the cyclic structure has lost its specific aldehyde or keton group.
But always the ring of the cyclic structure is closed with an O atom that has a position in between two C atoms.
This phenomena has consequencies for the reactivity of the substances.