Gas chromatography
Gas chromatography is a method that applies absorption.
Substances will be absorbed by a 'carrier'.
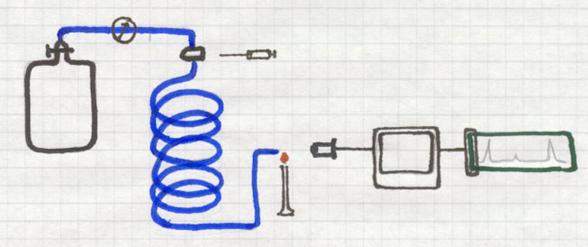
A long, thin, spiral shaped tube is full of a porous solid, the carrier.
Continuously an (inert) gas is passing that tube, or better: the carrier.
That gas could be, for example, He, coming from a cylinder where the gas pressure can be controlled very well. Helium is one, but there are other inert gases.
At the beginning of the tube there is an injection room where a gas mixture can be injected. That unknown gas mixture is to be investigated.
The injected small amount of gas mixture immediately is taken with the carrier gas He all through the tube and the solid carrier.
One component will move faster that the other, dependent on its absorbtion by the carrier. So one gas will leave the tube earlier than the other.
At the end of the tube, for example with a micro flame that react with color change, the outcoming component will change that color.
A detector can analyse that color as well as the intensity of the color, and give a signal that can be printed out.
Thus you cannot only determine which component was present in the mixture, but also its percentage.
