Disaccharides
Disaccharides are "dimers": built up of two monomers having a cyclic or a linear structure.
The bonding between these monomers could be called an 'oxygen bridge': you always will note the following bonding: C - O - C
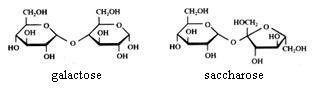
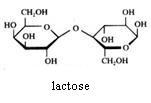
Maltose and lactose can be oxydised with Ag+ or with Cu2+.
Maltose also is built up of two monomers (2 x glucose), of which one can have a linear structure and the other only can have a cyclic structure.
The oxydisability of a cyclic structure depends on the position of the O (oxygen atom) in the ring.
If such a C - O bond in the ring can easily be opened (and the linear structure is made), than oxydation is possible.

