| CS2 (ΔE = ±0) | CO2 (ΔE = ±1.0) | H2O (ΔE = ±1.3) |
| Covalent bond | Covalent bond | Covalent bond |
| Non polar molecules | Non polar molecules | Polar molecules |
| there is no dipole | there is no dipole | Dipole |
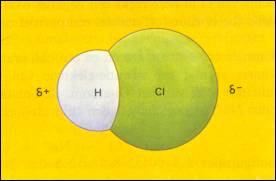
| There are no δ+ and δ- | There are δ+ and δ-; they overlap the centre points of each other |
The central points of δ+ and δ- do not overlap each other (remain at a distance) |
|
S=C=S
|
O = C = O
δ- δ+ δ- |
δ+
δ+
H H \ / O δ- |
